
Oxygen is referred to a diatomic molecule. Oxygen (O 2) is not a compound, however, because it has one single kind of atom (O). For example, carbon dioxide (CO 2) and sodium chloride (NaCl) are compounds because each one has two kinds of atoms that are bound by covalent or ionic bonds. CompoundĪ substance that is formed from more than one kind of atom chemically bound together. The gas law that combines Boyle's Law, Charles' Law, and Gay-Lussac's Law. Properties of a solution that depend on the number of solute molecules present but not on the identity of the solute. 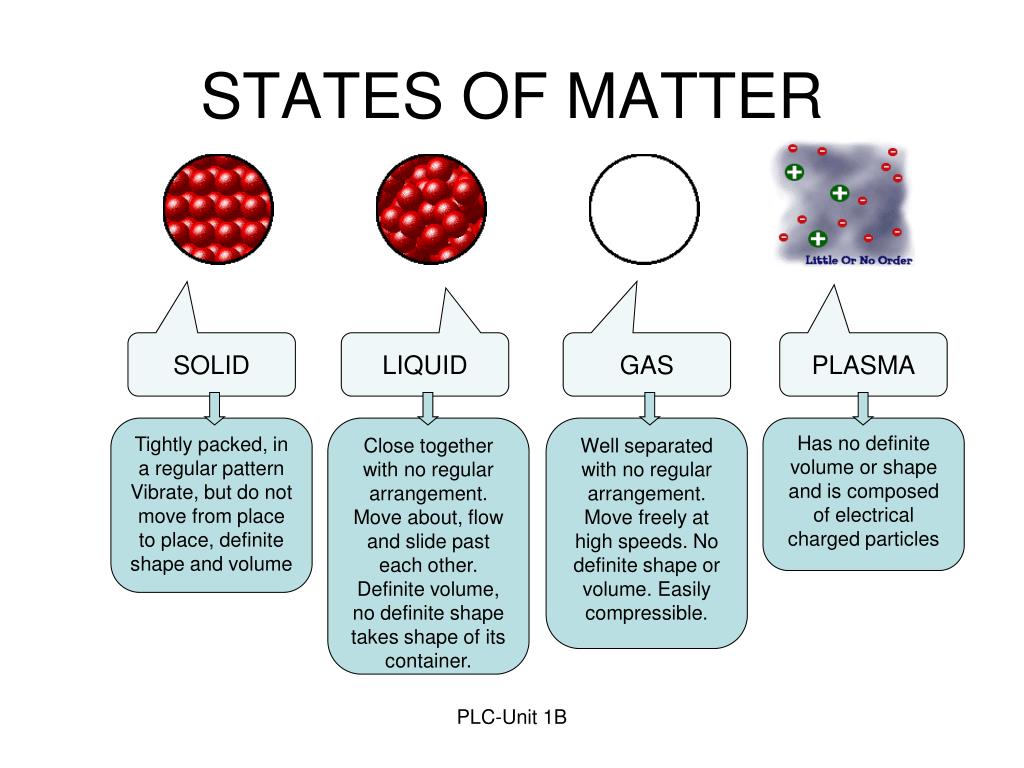
The intermolecular forces which cause a tendency in liquids to resist separation. Chemical ProcessĪ method of changing one or more chemicals or chemical compounds. The volume of a gas is directly proportional to its temperature if pressure and amount of gas remain constant. The movement of a liquid along the surface of a solid caused by the attraction of molecules of the liquid to the molecules of the solid. 
The pressure of an ideal gas is inversely proportional to its volume if the temperature and amount of gas is held constant. The boiling point of a solution is higher than the boiling point of the pure solvent (colligative property). The temperature at which the vapor pressure of a liquid is equal to the atmospheric pressure. AqueousĮqual volumes of an ideal gas contain equal numbers of molecules if both volumes are at the same temperature and pressure. Amorphous SolidsĪ solid that does not have a repeating, regular three-dimensional arrangement of particles. The attractive forces between unlike molecules. Solids, Liquids, and Gases Terms Adhesive Forces
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
